Lots of people say you have to use sulfate-free shampoo to avoid damaging or stripping your hair. There also people who say you have to use shampoo with sulfates to get your hair clean.
They are both wrong.
I’ve been paying more attention to hair science info on the internet lately. There are a lot of completely wrong explanations, and strong opinions, and people yelling at each other… but because I love pain apparently, I’m going to talk about it.
I’m starting with sulfate versus sulfate-free shampoos. I’ve talked about it a bit before, but I’m going into more depth this time. I think if we really dig into the chemistry of it, a lot of things about hair start making more sense – like why so many people can’t find the right products.
But before we talk about sulfates, we have to talk about the state of hair science – not just internet hair science, but actual academic hair science…
It’s a mess.
The video version is here, keep scrolling for the blog post version…
Hair is really diverse
There’s three big reasons I think there’s so much confusion around hair science.
Reason 1: Hair is really diverse – so much more so than skin. Different hair just does not respond to haircare products the same way.
Skincare seems complex, but when you break it down it’s actually pretty straightforward (maybe also it seems that way to me because I’ve been talking about it for so long).
With skin type there’s oily and dry and in between, then you have concerns like fine lines, pigmentation, acne. It takes a little while to understand your skin and find the right products for it (I have to plug my eBook because I keep forgetting to do that – it walks you through that process).
Related: The Lab Muffin Guide to Basic Skincare
But when it comes down to it, ingredients tend to work pretty similarly for everyone.
If a hundred people use glycerin on their skin, you might have 60 people’s skin get more hydrated and improve, 20 people’s skin stays around the same, the final 20 might get worse for some reason – like the glycerin helped some other ingredient penetrate and their skin got irritated, or they have an allergy. There’s still complexity, but you wouldn’t have half the people’s skin dry out more.
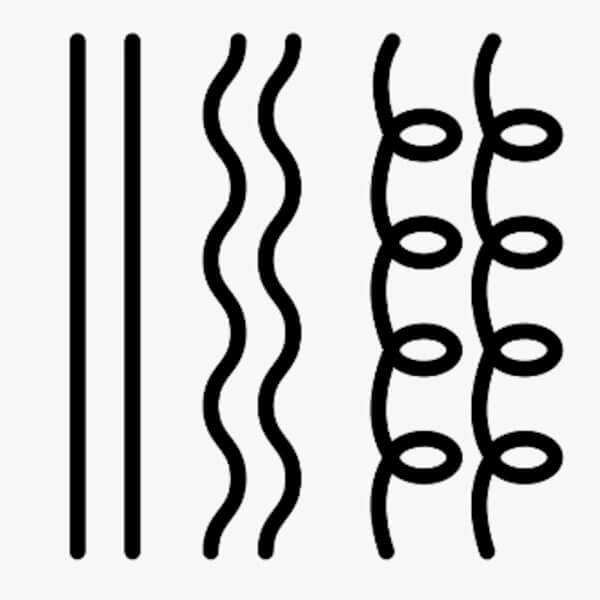
But that’s kind of how it is with hair. Even just with the shape of the strands – you have straight, curly, a million different levels and types of curly: kinky, tight coils, the coils might be rounder or flatter or a mix.
And shape makes a massive difference to how they respond to different products. Variation in geometrical shape is just not something we really have to think about that much with skin.
Wet versus dry detangling
Take wet versus dry detangling for example.
The way water interacts with hair is weird.-‘m not going to go into much detail here, that’s a whole topic for another 5 posts.
But in short: it weakens the bonds gluing the inside of the hair together (which is why hair gets stretchy when it’s wet), but adds stickiness to the outside of the hair, so hairs stick together more easily and you need more force to get the comb through.
Related Post: My Top (Mostly Cheap or Free) Haircare Hacks
Water also raises the cuticle scales. These are the roof tile-looking cells on the surface, and that means they can bash against each other more and chip.


So all of this means it’s less damaging to detangle hair when it’s dry – for straight hair.
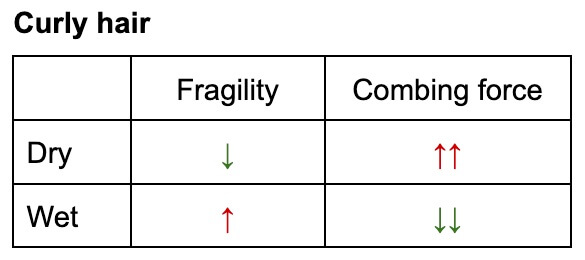
But it’s the opposite for curly hair. All of these effects still happen, but changing just the shape of the hair strand is enough to change the physics of how the hair interacts with a comb.
The cuticle still stands up more, the curly hair is still weaker on the inside and sticky on the outside, the hair is still more fragile.
But 2 big differences:
- Hairs stick to each other less because there’s less alignment (tessellation) between curly hair strands compared to straight hair. It’s like how spaghetti sticks together much more than spiral pasta.
- On top of that the weakening of the bonds inside the hair actually makes the curls loosen in shape. A looser curl means geometrically, there’s less chance of tangling and causing damage and breakage.
So the change in physics of how the curl interacts with the comb can offset the damage from manipulating wet, fragile hair to lead to less breakage – there’s a few studies showing this.
But where’s the line? Is it better to detangle wavy hair when it’s wet or dry? What if you use a brush versus a comb? Or a different kind of brush? What if you detangle from the bottom up? What happens if you half dry the hair?
That info just… doesn’t seem to exist.
And this is just water and hair shape. We’re not even talking about:
- other ingredients
- how often you wash your hair
- how much damage, the type of damage
- if you bleach or straighten your hair
- length
- oiliness…
There are a lot of variables.
Related Post: What to Expect After You Bleach Your Asian Hair
Which kind of overlaps with…
Hair science is really sparse
Reason 2: Hair science is really sparse. We just don’t know that much about hair.
I used to assume that hair was really straightforward. I mean, hair is dead, skin is alive, doing tests on hair should be really easy – buying a bunch of hair is so much easier than getting a bunch of volunteers.
But because of all this diversity with hair, each experiment just can’t necessarily be generalised.
On top of that there are tons of other variables that make a huge difference to hair, like what humidity the experiment started and ended with – that alone can massively change the results. And we live in places with different humidities, so it isn’t like if every study did experiments at 20% humidity it would solve the problem.
Hair scientists don’t even know the structure of hair in that much detail. The understanding is still evolving relatively quickly, and there just isn’t that much consistency with terminology even in the peer reviewed literature.
For example, around the late 90s, some hair scientists decided that half the protein in hair wouldn’t be called keratin anymore.
But not everyone got the memo, or maybe some hair scientists just didn’t agree – you’ll see things like “hair is 80% keratin by mass” in this paper from 2017.
Which isn’t a huge deal, because it’s clear they’re using the old definition – but if a paper says “we concluded that this ingredient works on keratin in hair”, what do they mean by “keratin”??
Hair products aren’t about individual ingredients
Reason 3: The two main hair products – shampoo and conditioner – the way they work isn’t about individual ingredients. The overall formulation is really important. And yes, we say that about skincare a lot, but it’s even more the case with haircare.
And this is central to the debate about sulfates in shampoos.
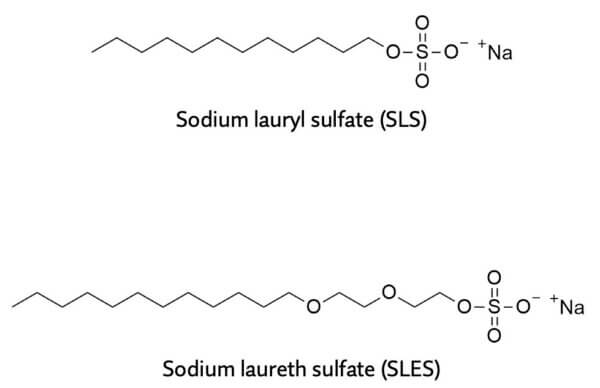
Sulfates usually refers to sodium lauryl sulfate and sodium laureth sulfate, two of the cleaning ingredients or surfactants found in cleaning products such as shampoos.
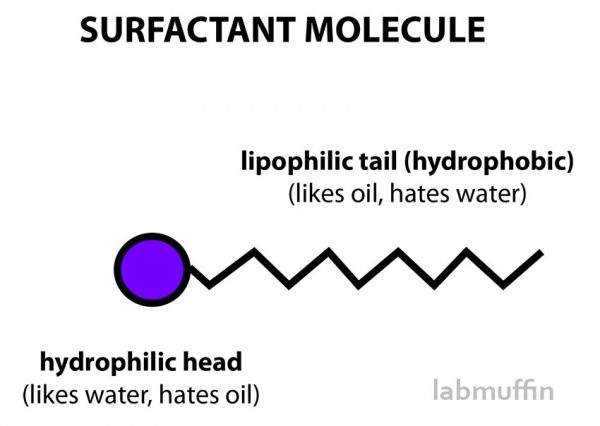
Surfactants are special because they have a head that likes water, and a tail that likes oil.
Oil and water don’t usually mix, so surfactants can help oil break up and mingle with water so you can rinse it away.
The tails are pretty similar in surfactants, so it’s really the heads we care about. Sulfates have sulfate head groups.
And yes, if we compare sulfates on their own with other surfactants on their own, they have really good cleaning ability. But because it’s hair science, there’s more complexity.
These 4 diagrams show 4 different ways that shampoos clean at a microscopic level – each diagram goes from left to right. The pink tadpoles are the surfactants.

You can see that the surfactants are working together to clean the hair. This is called a supramolecular process, where molecules are interacting without going through a chemical reaction. It’s like molecular choreography.
And all 4 of these are probably happening on your hair at the same time, to different extents – there’s probably more mechanisms still to be discovered.
How much of each one is happening depends on the formula of the shampoo – these pink tadpole surfactants won’t be the same, and they’ll have different tendencies to interact in different ways. But it also depends on what sort of stuff (“soils”) you’re cleaning off, because they’re also part of the choreography.
If you have bigger blobs of oil for example, then Figure 5 is probably happening more, where some of the oil breaks off multiple times instead of all of it coming off at once like in Figure 4. But if you have hair dye, then it might not break up and Figure 4 might be happening more.
Related Post: The Science of Hair Products: Shampoo and Conditioner
Figure 7 happens more at higher surfactant concentrations – which is another complex variable. It isn’t just about the concentration of surfactant in the shampoo formula. At the start of the shampooing process you’ve just put the shampoo on your hair so it’s concentrated, but if you add more water or start rinsing, then the surfactant concentration decreases and you might have less of this happening.
This is already pretty complicated, but it’s still a massive oversimplification of the process. Hair science will not give us a break!
If you look at the ingredients of almost any shampoo, it’ll have 3 or more different surfactants, which means the pink tadpoles in the diagram aren’t all the same. If you change the ratio of the ingredients or swap one of these surfactants with something else, the shampoo will work differently.
Plus there’s a bunch of other ingredients like polymers that aren’t shown in these diagrams that interact with the surfactants, and that’ll also affect how it cleans. And things like the texture of the shampoo, the opening of the bottle – that’s going to change how much you dispense, how it spreads on the hair.
Related post: Surfactants Are Everywhere, aka Stop Being Terrified of Chemicals
So you can see how complex it gets. And that means trying to predict what happens just by looking at whether some of these pink tadpoles are sulfates or not just doesn’t work – this single piece of information just doesn’t tell you much. It just isn’t as simple as it has sulfates so it’ll clean better, or it’ll strip or “degrease” your hair. If it was, it would be way easier for everyone to find the right shampoo.
Same with pH of shampoo. I see a lot of people saying you have to test the pH of your shampoo and again, it’s just not very meaningful when the whole situatiom is so complex. It’s like trying to predict how a food tastes from how yellow it is.
Even if you knew the full formula of a shampoo, it would be really difficult to predict how it’s going to interact with itself, let alone with different types of soils on hair at different stages of the shampooing process. It takes a lot of computational power to model a much simpler sort of system with far less moving parts than a shampoo.
This is why cosmetic formulators spend a lot of time just doing trial and error, making formulas and trying them out. Changing little things about the formula can make a huge difference.
Here’s an example: Good Housekeeping did a test with 10 shampoo and conditioner pairings. Some had sulfates and some didn’t, and there were salon and drugstore brands.
The set that stripped hair dye the least had sulfates – it was Tresemme’s Keratin Smooth Color set. They have an article where hair scientists talk about how complex shampoo formulation is. But then, even with their own results and two scientists with PhDs quoted, their article on colour-stripping shampoos still has this:
“Look for “sulfate-free” formulas. Sulfates sud the hair and cause pigment loss, says Bodt.”
These myths run deep.
Related Post: Busting Hair Conditioner Myths: Build-Up, Silicones, Weighing Hair Down etc.
How product design can mess things up
Despite all this, I think a lot of us have had the experience where we try a sulfate-free shampoo, and it does feel like it’s gentler or doesn’t clean enough (the way you frame it depends on whether your hair likes that). And the reason for this I think, is a kind of circular reasoning that happens quite a lot with consumer products.
Formulators know that people looking for sulfate-free probably want something that cleans less. They know there’s all these widespread myths about sulfates being harsh – they’ve been around since the 90s, like in this email quoted in Snopes’ 1999 article:
“SLS is used to scrub garage floors, and it is very strong”
Even if they don’t know that, it’s probably on the product brief, which is the description of the formula the brand has in mind that the formulator works off. It’s usually written by marketing people based on consumer research, and it’ll have the marketing claims that they plan to put on the product. Chances are the brief says “sulfate-free” and “gentle” or “not stripping”.
So there’s a good chance that if you grab a random shampoo with sulfates, and a random shampoo without sulfates, the one with sulfates will clean better.
Related Post: How Do Different Types of Hair Dye Work? The Science
It’s not because just adding sulfates automatically makes shampoos strip more – we saw it can be formulated to strip colour the least – it’s just because of product design. The formulator formulated with sulfates, and to clean hair more. They might have also added other ingredients to make the hair feel cleaner.
But brands don’t all have the same idea of what “gentle” is, and some brands choose to not follow this consumer trend, so there are going to be lots of exceptions.
It’s like how if you grab a shampoo in a black and green bottle, there’s a good chance it’ll smell minty, because product designers have decided that’s what men like.
This is the exact same situation with foam and cleansers. How well something foams doesn’t tell you how well it cleans. What’s happening on a molecular level is different.
How well something foams depends on how a formula interacts with air and water to stabilise a thin stretched out film of water. Cleaning is about how it interacts with oil and dirt.
Both cleaning and foam rely on surfactants, but there’s no real correlation between which surfactants foam best and which clean best. And there’s so much that depends on the overall formula (there are other non-cleaning ingredients that can be added to boost foam, like glycerin) – again, both are complicated supramolecular processes that involve lots of moving parts.
Related post: Are You Washing Your Face Wrong? Busting Cleanser Myths
But if something doesn’t foam when we use it, we tend to feel it isn’t cleaning well. It’s just a psychological thing, and I personally still have this gut feeling even though I know all of this – humans didn’t evolve to be perfectly rational in our feelings! So formulators make sure shampoos usually foam well for us.
But if we don’t see it, then it doesn’t have to foam. One example is detergents for dishwashers. They’re actually formulated kind of the opposite way. They need to clean well with as little foam as possible, because foam causes pressure and that can build up and make the door leak, and flood your kitchen.
Related Post: Hair frizz science: water and hydrogen bonds
Bottom line: Sulfate-free and sulfate-containing don’t really mean much. How well a shampoo cleans is too complicated to predict that easily.
It’s much more useful to look at what the shampoo is telling you. If it says “clarifying” it’s a shampoo designed to clean your hair better, regardless of whether it has sulfates or not, if it says “colour protection”, it can have sulfates and still strip dye less than a sulfate-free shampoo. If you stick to these rules about sulfate and sulfate-free, you would’ve missed out on the least colour stripping shampoo.
It’s also really useful to look at reviews from people with similar hair to you and try a sample before buying.
Let me know what you thought of this deep dive into hair science, what I should talk about next!
References
, , , , , Shampoo science: a review of the physiochemical processes behind the function of a shampoo. Macromol. Chem. Phys. 2023, 224, 2200420. doi:10.1002/macp.202200420
Zhou J, Ranjith PG, Wanniarachchi WAM, Different strategies of foam stabilization in the use of foam as a fracturing fluid, Adv Colloid Interface Sci. 2020, 276, 102104. doi:10.1016/j.cis.2020.102104
Saguin J, 11 Best shampoos for color-treated hair according to experts, Good Housekeeping, 11 March 2023 (accessed 11 May 2023).
Aral BK, What is sulfate-free shampoo — and do you actually need to use it?, Good Housekeeping, 5 Aug 2020 (accessed 11 May 2023).
Mikkelson D, Does sodium lauryl sulfate in shampoo pose a cancer risk? Snopes, 10 Jun 1999










This was very fascinating as usual, thank-you.
I was reminded of an instructional video on cleanliness I saw on YouTube from the 40’s a while ago. They said you had to scrub your hair with soap until when you rubbed your hair it ‘squeaked’. That was before conditioner was widely used I’m guessing and when they only washed their hair every two weeks or sometimes once a month.
So much of what we think of as clean or healthy when it comes to hair and skin is cultural and psychological! I couldn’t stand having ‘squeaky’ hair when wet, to me it would just mean I hadn’t put in enough conditioner.
This is so interesting! I love that you mention the insanely complicated nature of hair texture as well. I feel like we need to normalise the trial of hair products rather than relying on simplistic catch phrases. I wish more hair products came in 100ml bottles for trial. I got horrible irritation on my scalp recently from a shampoo I tried and had to throw out almost a whole 1L bottle, I hate waste.
From my personal experience with my wavy hair. I get the least amount of hair breakage when detangling with wet hair covered in conditioner and using a wide-tooth comb or a brush with very flexible bristles like a Wet Brush. Without the extra slip from the conditioner, I get breakage when detangling wet but I get the same amount when detangling dry as well as frizz and my waves become less wavy. Just in case you were interested in my experience. My hair is type 2b.